
Case Study
First-in-Class Development
— Selective DYRK1B Inhibitor
Highlights
With in less than 6 months, by designing no more than 20 molecules, excellent selectivity for the DYRK1B target was achieved.
Project Overview
Client
European Biotech Partner
Objective
To optimize the reference compound to achieve high selectivity ((Selectivity Index > 10) while maintaining high activity (IC₅₀ < 5 nM).
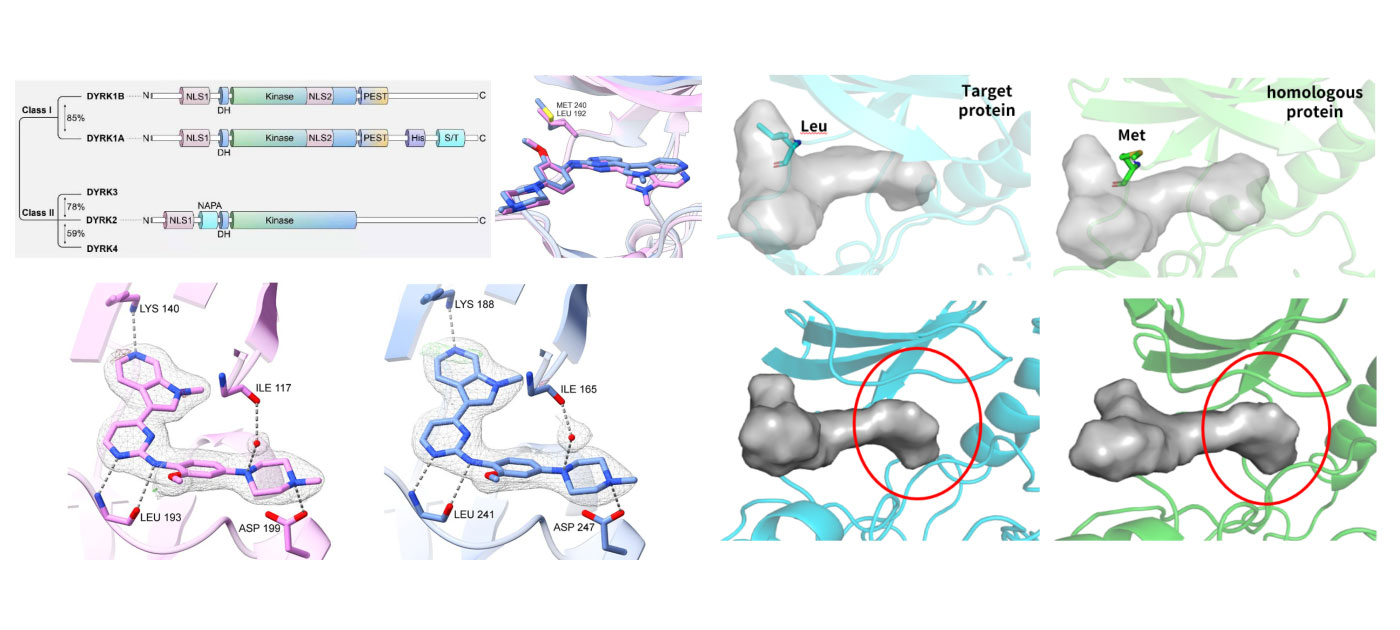
Main Challenge
DYRK1B shares 85% sequence identity with DYRK1A, and their binding pockets differ by only one residue in the hinge region. Crystal structures revealed no significant conformational differences.
Design Strategy /
Approach
Approach
- Divamics applied multi-scale molecular dynamics (MD) simulation to analyze pocket flexibility and guide rational ligand optimization.
- By exploring the conformational behavior of the hinge residue (Met → Leu), hydrophobic extensions were introduced to strengthen Leu-specific interactions.
- Additional pocket space on the right-side region was leveraged to further enhance selectivity.
Key Results /
Milestones
Milestones
- 20 compounds were designed and synthesized within six months.
- The optimized compound achieved enzyme activity below 5 nM and >10-fold selectivity for DYRK1B over DYRK1A.
- MD analysis confirmed a stable hydrophobic interaction pattern that explained the enhanced selectivity.
Interested in how Divamics can accelerate
your next discovery project?
Contact Us

